SIL 50mg + TAD 20mg Blend – 50 Tablets
- Estimated Delivery : Up to 4 business days
- Free Shipping : On all orders over $300
- 3rd Party Tested Premium Peptides : 99% Purity
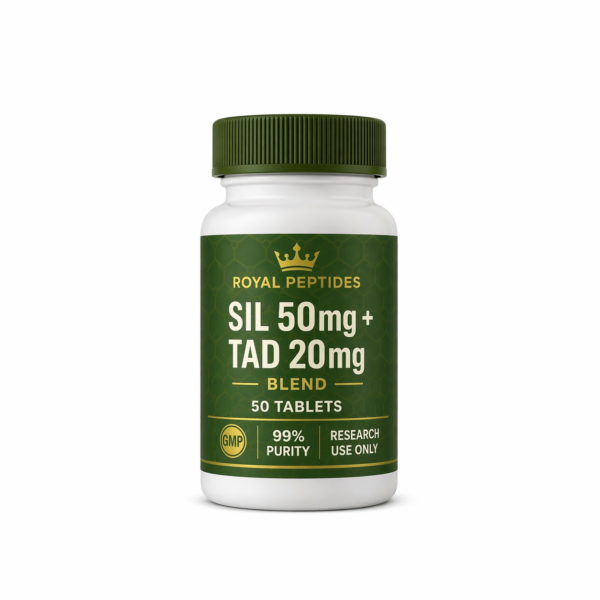
SIL 50mg + TAD 20mg Blend – 50 Tablets | Royal Peptides
A premium dual-compound tablet formulation combining SIL (50mg) and TAD (20mg) in a precise, easy-to-use format. Each tablet is manufactured for consistency and accuracy, delivering reliable dosing across every batch for advanced research applications.
Produced in GMP-compliant facilities and subjected to strict quality control standards, this blend maintains exceptional purity, stability, and performance.
Key Features:
50mg SIL + 20mg TAD per tablet
50 tablets per bottle
99%+ purity standard
Dual-compound blend for advanced research
GMP-certified manufacturing
Consistent, pre-measured tablet format
Research Use Only
Designed for researchers seeking a convenient, high-quality combination formula with dependable dosing and trusted sourcing.
New To Peptides?
Review the essentials before placing an order. This section helps buyers understand product format, storage expectations, documentation, and key details to verify before purchase.
Product Format
Most compounds are supplied in lyophilized powder form rather than as pre-mixed solutions.
Storage Expectations
Proper storage conditions are important for maintaining compound stability and quality.
Check Product Details
Confirm compound name, vial size, total amount, and blend composition where applicable.
Review Documentation
Read the product description and any available testing or quality documents before ordering.
Educational References
Glossaries, calculators, storage notes, and general reference materials can reduce confusion.
No Instructions Provided
We do not provide dosage recommendations, administration instructions, or reconstitution guidance.
Storage Instructions
Maintain proper storage conditions to preserve product integrity before and after reconstitution.
Research Use Only. This product is not intended for human or animal use.